Research areas with patient participation.
The lab's translational work spans implantable BCIs, deep brain stimulation, and stereo-EEG-guided neuromodulation. The areas below describe current and recent published work; potential participants and referring physicians can contact the lab directly to inquire about enrollment.
Specific eligibility, study status, and IRB protocol numbers vary. The fastest route to current information is direct email. For a participant-facing overview of how research participation works (consent process, what to expect at the OR, healthy controls), see Research participation.
Currently enrolling and ongoing.
Three studies are currently open to patient participation. Email the lab to inquire about specific eligibility and enrollment.
Cuneiform nucleus DBS for freezing of gait in Parkinson's disease

Sensing-Enabled Directional Deep Brain Stimulation of the Cuneiform Nucleus for Levodopa-Resistant Freezing of Gait in Parkinson's Disease
A prospective, multicenter, double-blind, crossover phase 1/2 trial evaluating the safety, feasibility, and preliminary efficacy of directional DBS targeting the cuneiform nucleus (CnF) for treating levodopa-refractory freezing of gait (FOG) in Parkinson's disease. Eighteen participants (six per site) will be enrolled across the University of Pennsylvania (coordinating center), UCSF, and the University of Miami.
Participants undergo bilateral directional DBS lead implantation in the CnF using tractography-based targeting and intraoperative microelectrode recordings. After recovery, participants are randomized in a double-blind fashion to active stimulation or sham for two months, then cross over to open-label stimulation for the remainder of the 6-month follow-up. The sensing-enabled platform (Medtronic Percept PC/RC) records local field potentials in the operating room and during walking tasks, supporting development of biomarker-informed adaptive DBS.
Primary outcomes: Timed Up and Go (TUG) and Freezing of Gait Questionnaire (FOGQ). Secondary outcomes include pirouette test times, PDQ-39, PDQL, MDS-UPDRS motor subscores, and adverse event rate. Exploratory outcomes include LFP-based FOG biomarkers and cognitive-loading and anxiety challenge paradigms.
Builds on a completed FDA IDE-approved pilot study (n=4) at Miami in which all patients showed marked improvements in objective gait measures, including a mean TUG reduction of 45.5 seconds (p = 0.032) and a 27.5% mean reduction in FOGQ scores (p = 0.0132). Directional stimulation was well-tolerated with no permanent device-related serious adverse events.
Intraoperative electrophysiological recordings during awake brain surgery
Intraoperative Electrophysiological Recordings and Non-Invasive Wearable Sensors for Movement Capture During Awake Brain Surgery
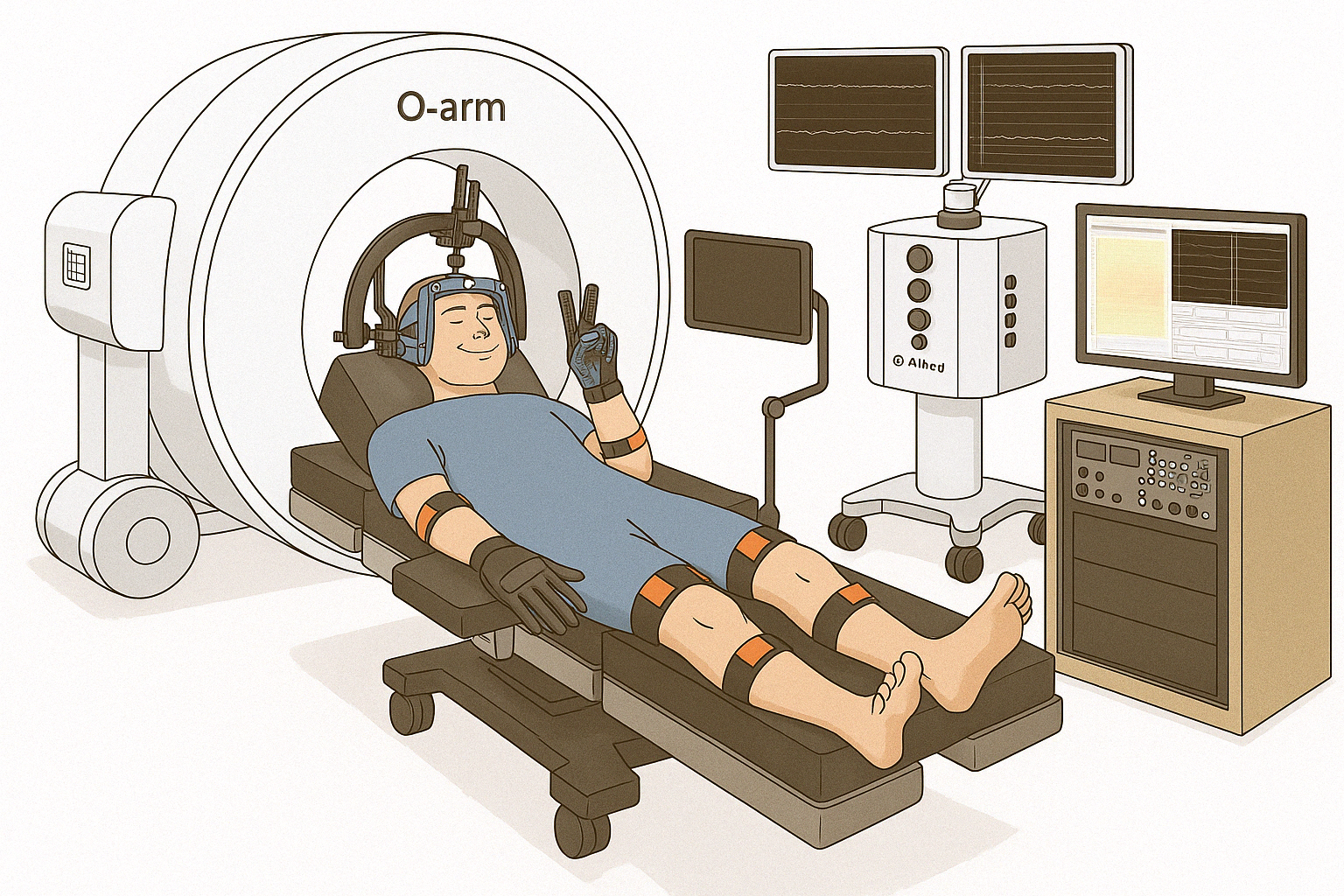
This study takes advantage of the rare access to the human brain afforded during awake brain surgery to understand how the brain controls movements. We use non-invasive wearable sensors to measure movement while recording from the brain surface and, where deep brain electrodes are already being placed for clinical care, from deep brain structures. Knowledge gained will be of benefit to understanding brain function and to the development of treatments for patients with paralysis using new brain-computer interface technologies.
Who can participate
Adults aged 18 to 75 already scheduled to undergo awake craniotomy or burr hole procedures that expose the cerebral cortex — including deep brain stimulation, supratentorial craniotomy for tumor, epilepsy, or cerebrovascular disorders. Participants undergoing DBS surgery must have no prior history of seizures.
What participation involves
Participation occurs during a portion of the surgery already planned for clinical reasons. For DBS patients, the long-term clinical electrode is placed deep in the brain as planned for the patient's therapy. Just before the patient is woken up, a research electrode array (approximately 2 cm × 2 cm, with 1,024 contacts — hundreds of times higher resolution than commonly available electrodes) is placed on the surface of the brain. Cortical and subcortical recordings are then collected during simple movement, language, or perceptual tasks while the participant wears motion-capture gloves that reconstruct hand and arm movement in 3D, synchronized with the neural recordings. Small electrical pulses may be delivered to the arms, hands, or fingers while under anesthesia.
Active participation lasts approximately 15–20 minutes during surgery. Setup takes about 25 minutes and is performed in parallel with surgery, so it does not lengthen the procedure. Total participation is under 50 minutes. There are no follow-up research visits, and no direct benefit to participants.
Penn Medicine produced a short video that scrubs into the OR and tours the dry lab. Watch on YouTube → "Thought-Controlled Prosthetics: A Brain-Computer Interface Breakthrough" (4 min · Penn Medicine, 2024).
Study aims
- Evaluate the effectiveness of various electrode arrays for recording cortical activity.
- Evaluate how electrophysiological signals in the sensorimotor cortex and subcortex relate to natural behaviors and stimuli.
- Evaluate how cortical and subcortical activity correlates with performance on simple tasks.
- Evaluate how cortical and subcortical activity relates to discriminability of artificially elicited percepts.
Where
Pennsylvania Hospital, Department of Neurosurgery. Five-year study, target enrollment 75 subjects.
Spinal cord recordings during SCS implant or trial
Intraoperative and Postoperative Electrophysiological Epidural Spinal Recordings with Simultaneous Non-Invasive Wearable Sensors for Movement Capture
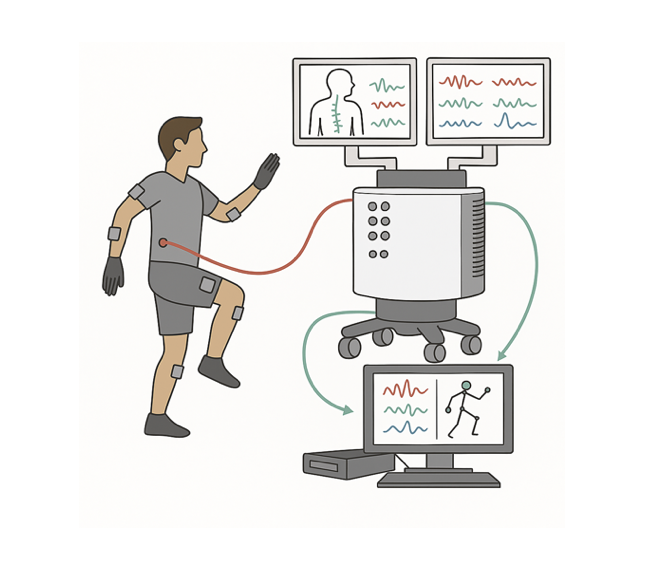
This study takes advantage of the rare access to the human spinal cord afforded by spinal cord stimulator (SCS) implant surgery and externalized percutaneous trial leads to understand how movement signals are represented in the cord. Special wearable sensors measure body movement while electrode arrays — including custom-fabricated high-density Ti3C2 MXene arrays developed in collaboration with the Vitale Lab — record from the epidural space. Knowledge gained will benefit understanding of spinal cord function and inform development of next-generation BCIs for patients with paralysis.
Who can participate
Adults 18–75 already scheduled for an SCS implant or percutaneous trial in the thoracic or cervical cord — typically for treatment of refractory chronic pain syndromes (complex regional pain, failed back syndrome, peripheral neuropathy, radiculopathy). Both intraoperative recordings during implant and postoperative recordings in patients with externalized trial leads are part of the protocol.
What participation involves
Intraoperative arm: a custom-fabricated high-density MXene recording array is temporarily inserted into the epidural space during the patient's already-scheduled SCS implant. Under light anesthesia, simultaneous epidural recordings and limb-movement kinematics are captured while the patient performs single-joint and complex multi-joint movements. Total research time is approximately 45–50 minutes including setup; the active recording portion is 15–20 minutes. The surgical approach and amount of epidural access are not altered to accommodate the research, and array placement attempts are limited to two with a hard stop if there is any safety concern.
Postoperative arm: for patients with external access to SCS leads (typically trial percutaneous SCS), the leads are connected to research recording hardware in the lab while the patient performs about three hours of movement and sensory tasks. The stimulator is reconnected to its original power source at the end of the session.
Motion capture uses the Stretchsense MoCap Pro glove and an Xsens MTw Awinda inertial sensor system, yielding a 74-degree-of-freedom skeletal model tracked in real time. Time-synchronized video and audio enable offline alignment of neural and behavioral data.
Study aims
- Evaluate the effectiveness of various electrode arrays — including high-density MXene arrays — for recording epidural spinal cord activity.
- Evaluate how spinal cord electrophysiological signals are related to natural movements and sensory stimuli, intra- and post-operatively.
- Evaluate how spinal cord activity correlates with performance on simple tasks.
- Evaluate how spinal cord activity correlates with complex tasks like walking, post-operatively.
Where
Pennsylvania Hospital, Penn Medicine Spine Center, and the Hospital of the University of Pennsylvania. Five-year study, target enrollment 50 subjects (up to 10/year).
Foundational work the current program builds on.
The lab's current translational research at Penn builds on prior work led by Dr. Cajigas at the University of Miami and during fellowship at UCSF. Two completed lines of work are summarized below; full publication lists are on the Publications page.
Implantable brain-computer interface for spinal cord injury
Fully implanted ECoG-based BCI for hand grasp and stepping
Long-term work in a 21-year-old male subject with C5 ASIA A cervical quadriplegia using a fully implanted, portable BCI. The subject demonstrated volitional control of hand grasp via functional electrical stimulation and a mechanical orthosis, and BCI-controlled stepping via upper-limb motor imagery. The line of work was followed for five years in the home environment, with stable decoder performance throughout.
Key publications
Cerebellar DBS for movement disorders in cerebral palsy
Cerebellar deep brain stimulation in cerebral palsy
Early results suggest cerebellar DBS may benefit patients with movement disorders due to cerebral palsy, particularly dyskinetic CP. Published work outlines the rationale and looks ahead to a larger clinical trial led by collaborators at UCSF.
Inquiries about participation or referral
- Patients and families: see For Patients & Families for plain-language information, then email the lab.
- Referring physicians: contact Penn Neurosurgery to refer to Dr. Cajigas (Functional & Stereotactic Service).
- Direct: iahn.cajigas@pennmedicine.upenn.edu

